Department of infection Sciences
The Department of Infection Sciences, incorporating Bacteriology, Serology and Molecular Biology, is situated within the Main Building of The Royal Stoke University Hospital (RSUH). The Department is a large modern laboratory that processes approximately 1.5 million tests per year. It is part of the Pathology Directorate and North Midlands and Cheshire Pathology Service (NMCPS) Division of the University Hospitals of North Midlands NHS Trust, providing the infection diagnostic service for the North Midlands and Cheshire Pathology Service partnership.


The Department provides a comprehensive, service for the University Hospitals of North Midlands, East Cheshire NHS Trust, Mid Cheshire Hospitals NHS Foundation Trust, and the local health economy including the Combined Health Care Trust, Primary Care Trusts and surrounding General Practitioners. The laboratory also works in partnership with the United Kingdom Health and Security Agency (UK HSA) in Birmingham and the Environmental Health Departments within Staffordshire in the management of outbreaks.
The Department provides a comprehensive, UKAS accredited (ISO 15189) Infection Science Diagnostic service as part of the North Midlands and Cheshire Pathology Services.
Sample containers
All samples should be submitted in appropriate containers – refer to individual tests for details. It is the responsibility of the service user to ensure that sample containers are stored correctly in an appropriate environment. Please refer to the manufacturers’ guidance for details.
Specimens
All specimens must be labelled with the following:-
- Surname/forename or other unique patient identifier
- Date of birth
- Sender’s sample number
- Date of collection of specimen
Printed specimen labels should be used wherever possible. Please note that unlabelled specimens cannot be processed and may be discarded.
Request forms
Most requests are done electronically. The following applies only to the manual requesting method.
Confirm the identity of the patient.
The patient’s name and ward (or location) must be on all specimen containers.
A request card must accompany all specimens to the laboratory. This should show clearly the patient’s details including:
- Name
- Unit number/NHS number
- Age (date of birth preferred)
- Ward/GP name and number
- Type of specimen
- Date and time of sample
- Tests required
- All relevant clinical data including any antibiotic treatment
- Risk status if applicable (legal requirement)
If either the request form is not completed correctly or the sample is not adequately labelled a request may be rejected as processing the samples may result in mis-identification of the patient constituting a clinical risk.
Preparation of the Patient
Ensure that the patient is fully informed as to the nature of the investigation to be performed and that their consent is obtained. They should understand what the procedure involves, why they need it and what the consequences may be if the investigation is not performed.
Where a patient has to go away and obtain a sample themselves they should be fully instructed in the procedure for obtaining that specimen.
Collection of Specific Specimen Types
The GP or healthcare professional (such as a nurse) has a responsibility to explain to the patient how to collect a clinical sample if they will be doing this themselves without direct supervision. The details for specific types of sample that may be collected by patients themselves are below.
Urine Specimens for Microscopy and Culture
A urine sample can be collected at any time of day. However, you should not excess water to produce urine as this will make the sample too dilute and it may be rejected by the laboratory.
Usually, for microbiological examination, a midstream urine (MSU) is collected. This means that the first or last part of urine that is passed is not collected. This reduces the risk of the sample being contaminated with bacteria from within or around the urethra or from the hands.
- Urine must be collected into a completely clean and sterile container. Failure to do so may result in a misleading result and the specimen may be rejected.
- The container should be one with a red top as specified by the laboratory. This has boric acid crystals in as a preservative.
- If the sample is not transported to the lab straight away it should be stored in a fridge, in a sealed plastic bag,
To collect a clean urine sample:
- Label the container with the patient’s name, date of birth and the date
- Wash the hands
- Start to urinate, but don’t collect the first part of urine that comes out
- Collect a sample of urine "mid-stream" in the sterile screw-top container
- Screw the lid of the container shut
- Wash the hands thoroughly
Urine for Pregnancy Test
A urine sample for a pregnancy test should be collected as for a culture except:
- The sample should ideally be the first urine passed in the morning, known as an “Early Morning Urine” (EMU).
- The sample should be in a white topped container with no boric acid
- The sample does not need to be a midstream sample
Faeces Specimens for all Microbiology Tests
- The stool sample should be collected in a clean, dry screw-top container. This should be a laboratory specified container and should be provided to the patient by the doctor or healthcare professional. However any clean container can be used as long as you can seal it.
- The container should be labelled with the patient’s name, date of birth and the date
- Place something in the toilet to catch the stool, such as a potty or an empty plastic food container, or spread clean newspaper or plastic wrap over the rim of the toilet
- Make sure the sample doesn’t touch the inside of the toilet
Try not to collect urine or water (from the toilet) with the stool sample but don’t worry if this happens.
If you need to urinate, do this first, and then collect the stool sample. - Use the spoon or spatula that comes with the container to place the sample in a clean screw-top container and screw the lid shut.
For a laboratory contained aim to fill around a third of it, about the size of a walnut. - Put anything you used to collect the sample in a plastic bag, tie it up and put it the bin
- Wash your hands thoroughly with soap and warm running water
- If there is a delay in delivering the sample the container should be put in a fridge in a sealed plastic bag until it can be delivered to the lab
Sputum Specimens for all Microbiology Tests
Good quality sputum samples are essential for accurate microbiological diagnosis of pneumonia but also acute tracheitis and bronchitis. Samples contaminated with saliva are difficult to interpret and can be misleading.
The most common way is for you to cough a sample into a sterile specimen pot, which your GP or healthcare provider should provide.
Sputum sample size should be 5 – 10 ml where possible and preferably early morning specimens.
NB For tuberculosis we require 3 specimens which must be collected at least 24 hours apart, preferably on consecutive days.
- Drink plenty of fluid the evening before to help loosen the phlegm
- Do the sample as soon as you wake up in the morning, preferably before drinking anything or having breakfast, as this may confuse the results. This is particularly important if the sample is to test for possible tuberculosis (TB).
- Do not clean your teeth or use mouthwash prior to doing the test as this may kill any bacteria in the sputum.
- Take three deep breaths to loosen the secretions (if you have a nebuliser you may want to use this first) and hold the air in for five seconds.
- Take another deep breath and cough hard until some sputum comes up into your mouth then force out a deep cough and bring the sputum up into your mouth.
- Spit the sample into the collection container.
- If you are unable to bring up much sputum (minimum is about the same size as your little finger nail) on the first attempt then try again until you have enough.
- Screw the cap on the cup tightly so it doesn’t leak
- Take the sample to your GP surgery, clinic or to the laboratory on the same day if possible.
- The container MUST be labelled with the patient’s name, date of birth and the date
NB Compliance with postal, transport and storage regulations is essential, for postal samples use only the containers and packaging provided.
Transportation of Specimens
Specimens should be taken into sterile, leak proof containers supplied by the laboratory.
The specimen should be sealed in the bag and the attached card completed, with full patient information, clinical history and specimen details as appropriate.
The specimen should be transported to the laboratory as rapidly as possible after collection to allow the most accurate interpretation of results.
Specimens sent by post or by courier must be in a sealed container, surrounded by sufficient absorbent packing material to take up any leakage in the event of damage during transit, sealed in a plastic bag and placed in an approved outer container which meets current postal or other transport regulations.
Contact the Microbiology department for further information.
Guidance on the transport of infectious substances (including links to current European agreements and information from the HSE) may be found on the following web page:
http://www.dft.gov.uk/dangerous goods
Procedure for “High-Risk” Specimens Category 3
High risk specimens are those that potentially contain category 3 pathogens. For a list of organism categories refer to the Advisory Committee on Dangerous Pathogens (ACDP) Approved List of Biological Agents
High-risk specimens must be labelled with the appropriate sticker
The accompanying request card must also carry the high risk label, as well as sufficient clinical data to enable experienced laboratory staff to know what special precautions or additional tests are required.
See individual tests for specific details.
Procedure for ‘Major Pathogens’ Specimens Category 4
Special arrangements are required for the collection and transportation of specimens involving suspected hazard group 4 agents – contact the Consultant Microbiologist via the phone numbers above or by bleep via the hospital switchboard BEFORE taking any samples.
Clinical Data:
It is essential that appropriate clinical information is supplied. This will include the specific anatomical site and the nature of the sample, history of any foreign travel, contact with animals and occupation if this is relevant. Failure to provide relevant information may mean that the most appropriate investigation is not performed.
Bacteriological Investigations
Delay in transport:
This may affect the viability of pathogens and allow overgrowth of normal flora or contaminating organisms. Morphological appearance of cells may also be affected
Excessive temperature:
Generally bacteriology samples should be kept cool. Bacterial activity may also increase leading to misleadingly high counts for pathogens or overgrowth of normal or contaminating flora. Excessively high temperature may kill the target organism. NB Some organisms do not tolerate cooling well and so there is no substitute for rapid transport.
Inappropriate specimen site or transport medium
Optimal recovery of the target organisms will not be possible
Antibiotic treatment:
Current or recent treatment with antibiotics should be provided as this may affect the culture results obtained.
Helicobacter faecal antigen testing:
False negatives can occur if antimicrobials, proton pump inhibitors or bismuth preparations are administered in the 2 weeks prior to sampling.
Virology, Serology and Molecular Microbiology
Serology tests:
Samples which are highly haemolysed, hyperlipaemic or which contain microbial contamination should not be sent. Heat inactivated samples may give rise to erroneous results in a number of assays and should not be sent. Serum or plasma samples should be stored at 2-8°C for no longer than 7 days – if stored for a longer period of time, they should be frozen at -20°C or lower. Repeated freeze-thaw cycles should be avoided, as this may degrade the analyte sought and cause inaccurate quantitation or false negative results. If sending samples at ambient temperature, transit time must be less than 72 hours. Please note that while post-mortem samples may be accepted, only a limited number of tests have been evaluated for use with these samples.
Certain assays require serum only – plasma samples are not suitable. Specific requirements are listed in the individual tests. When sending paired sera, please ensure samples are taken at least 14 days apart.
Molecular tests:
Serum/plasma
EDTA plasma is preferable to serum, as degradation of nucleic acid can occur in serum/ clotted samples, which may result in under-quantification of viral load. Samples should be sent as soon as possible, or frozen at -20°C or lower. Repeated freeze-thaw cycles (>3x) may result in under-reporting and should be avoided. Samples which are highly haemolysed, hyperlipaemic or which contain gross microbial contamination should not be sent. Heparinised samples or samples from patients who have received heparin, may give erroneous results and must not be sent – please contact laboratory for advice.
Other samples
Do not send dry swabs, charcoal swabs, swabs in bacterial transport gel or swabs with wooden shafts, as all are unsuitable for molecular testing.
Details of any antiviral therapy should be given wherever possible.
Whole (unseparated) blood samples:
Certain tests (e.g. HIV and HTLV proviral DNA) require whole unseparated blood collected on EDTA. Samples should be sent to the laboratory as soon as possible after collection. Where possible, whole blood samples should not be sent over a weekend. Samples over three days old may not be suitable for testing.
Tissue samples:
Tissue samples are not routinely tested by PCR at this laboratory but may be referred to a reference laboratory. Please contact laboratory before taking tissue samples for PCR as they may not be validated.
CSF, oral fluid, urine and other samples:
Samples should be sent in the appropriate media for the test requested – see individual tests
Samples in incorrect media may give rise to erroneous results and will not be tested.
Measurement Uncertainty in Microbiology
This is a parameter, associated with the result of a measurement that defines the range of the values that could reasonably be attributed to the measured quantity. Measurement Uncertainty is performed throughout the department and is available on request.
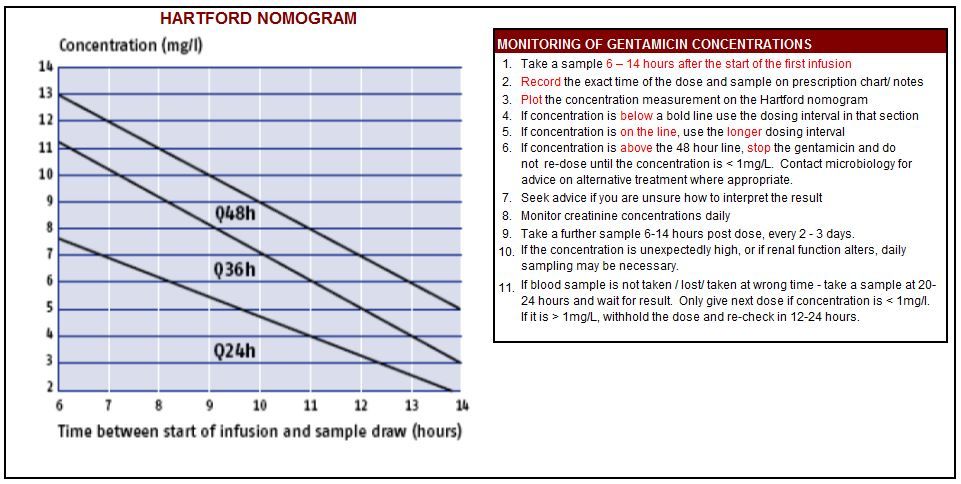
A copy of the Gentamicin calculator (Hartford Regimen) is shown below
Monday to Friday:
Available via departmental pager: 8.00am to 9.00am
Available via telephone (01782 674898, ext 74898): 9.00am to 5.00pm
Available via departmental pager: 5.00pm to 7.30pm
Overnight cover via departmental pager: 7.30pm to 8.00am
Saturday:
Available via departmental pager: 8.00am to 9.00am
Available via telephone (01782 674898, ext 74898): 9.00am to 1.00pm
Available via departmental pager: 1.00pm to 5.00pm
Overnight cover via departmental pager: 5.00pm to 8.00am
Sunday:
Available via departmental pager: 8.00am to 5.00pm
Overnight cover via departmental pager: 5.00pm to 8.00am
Serology: Monday to Friday:
Available via telephone (01782 674941, ext 74941): 8.30am to 5.00pm
Serology: Saturday & Sunday:
Available via telephone(01782 674941, ext 74941): 8.30am to 4.00pm*
* Skeleton staff only, please contact Bacteriology secondary if unable to contact virology during these times.
Molecular laboratory: 24/7
Available via telephone (01782 674960, ext 74960)
Biomedical Scientist cover 9am to 8pm 7 days
